Present in waters and wastewaters, surfactants are discharged via aqueous waste from households, industrial laundering, and other cleansing operations. Generally present in detergents and other cleaning agents, a surfactant molecule consists of a strongly hydrophobic group and a strongly hydrophilic group, permitting solubility in both aqueous and non-aqueous media. When the hydrophilic group is negatively charged, it is deemed an anionic surfactant; when the hydrophilic group is positively charged, it is deemed a cationic surfactant.
The HI97769 uses an adaptation of the USEPA method 425.1 and the Anionic Surfactant as MBAS method per Standard Methods for the Examination of Water and Wastewater to measure anionic surfactant concentrations of less than 3.50 mg/L (ppm) (as sodium dodecylbenzenesulfonate [SDBS]). When the methylene blue reagent is added to samples containing anionic surfactants, the sample will turn a blue hue; the greater the concentration, the deeper the color. The associated color change is then colorimetrically analyzed according to the Beer-Lambert Law. This principle states that light is absorbed by a complementary color, and the emitted radiation is dependent upon concentration. For anionic surfactant determination, a narrow band interference filter at 610 nm allows only orange light to be detected by the silicon photodetector and omits all other visible light emitted from the LED. As the change in color of the reacted sample increases, absorbance of the specific wavelength of light also increases, while transmittance decreases. Using a pre-programmed curve a result is then displayed.
The HI9776 has an innovative optical system that offers superior performance in accuracy, repeatability, and the short amount of time it takes to perform a measurement. This compact, waterproof meter is extremely user friendly with a tutorial mode that walks the user graphically, step by step, in performing a measurement. The use of a backlit dot matrix LCD allows the use of virtual keys which makes the operation of the meter very intuitive including selecting different units of measure, reviewing GLP data, recalling the last 50 measurements, and customizing the meter to user preferences.
HI97769 is completely waterproof including the cuvette holder that is designed with ridges to protect the optical path from getting scratched by the cuvette and a gasketed battery compartment that holds three common AA batteries. The compact design fits comfortably in the hand for use in the field or on a table for benchtop use. The LCD is backlit for easy viewing under all conditions.
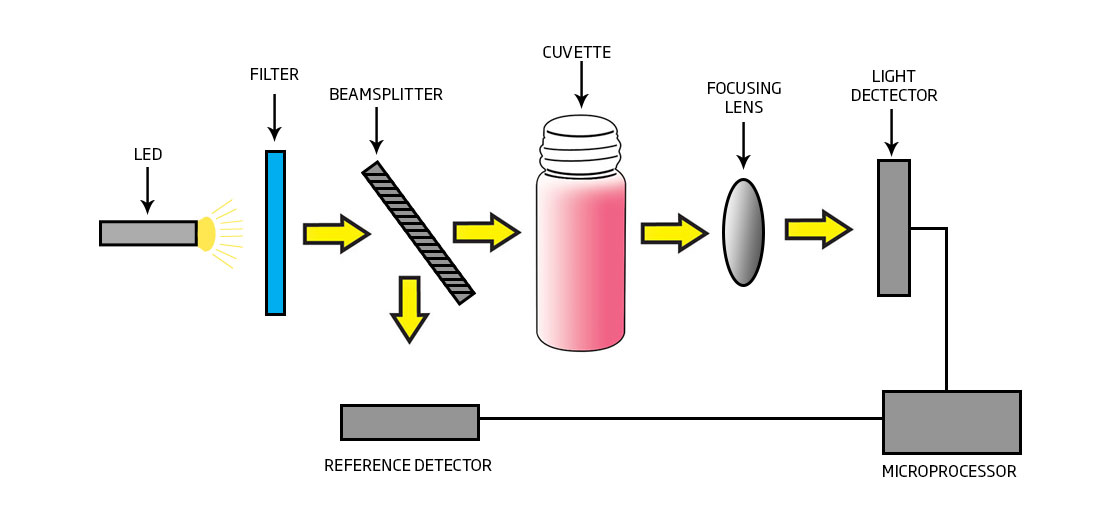
- LED that generates very little heat.
- 8 nm narrowband interference filter that is accurate to ±1 nm.
- Reference detector that modulates the voltage to LED for consistent light output.
- A concave focusing lens that reduces errors from imperfections in the cuvette.
On-Screen Features
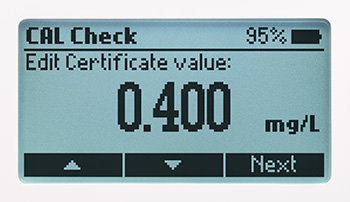
CAL Check
Advanced features including CAL-Check to verify performance and if necessary, recalibrate.
Setup Options
Setup options for meter personalization include date and time format, language, and enabling the tutorial mode.
Backlit Display
Backlit dot matrix LCD that offers an exceptionally intuitive user interface that is easy to read and understand.

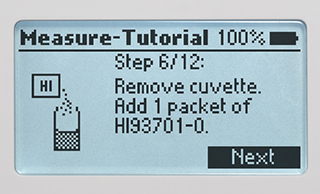
Tutorial Mode
Tutorial mode for step-by-step instructions to guide a first-time user in how to perform a measurement correctly.

Reaction Timer
Built-in reaction timer that ensures consistency amongst multiple users.
HI97769 FEATURES/BENEFITS:
Stable Light Source:
- The internal reference system of the HI97769 photometer compensates for any drifts due to power fluctuations or ambient temperature changes. With a stable source of light the readings are fast and stable between your blank (zero) measurement and sample measurement.
High Efficiency Light Source:
- LED light sources offer superior performance compared to tungsten lamps. LEDs have a much higher luminous efficiency, providing more light while using less power. They also produce little heat, which could otherwise affect electronic stability.
High Quality Filters:
- Improved optical filters ensure greater wavelength accuracy and allow a brighter, stronger signal to be received. The end result is higher measurement stability and less wavelength error.
Greater Light Yield:
- A focusing lens collects all of the light that exits the cuvette, eliminating errors from imperfections and scratches that may be present in the glass. The use of the convex lens reduces the need for indexing cuvettes.
CAL Check Functionality:
- Hanna’s exclusive CAL Check feature allows for performance verification and calibration of the meter using NIST traceable standards. Our CAL Check standard vials are developed to simulate a specific absorbance value at each wavelength to verify the accuracy of subsequent readings.
Large Cuvette Size:
- The sample cell of the HI97769 fits a round, glass cuvette with a 25 mm path length. The relatively long path length of the sample cuvette allows the light to pass through more of the sample solution, ensuring accurate measurements even in low absorbance samples.
Intuitive Dot Matrix Display:
- The HI97769 is designed with a backlit, graphic LCD. With virtual keys, a battery status indicator, and error messages. Users will find the meter interface intuitive and easy to read. A dedicated help key provides information relating to the current meter operation, and can be used at any stage in the setup or measurement process to show contextual help.
Auto-off Protection:
- The meter uses three common AA batteries that allow for about 800 measurements to be taken. The auto-off feature automatically shuts off the meter after 15 minutes of inactivity in order conserve battery life.
HI97769C is supplied with CAL-Check standards, cuvettes (2), caps (2), plastic inserts for cuvette, (2), scissors, cuvette cleaning cloth, 1.5V AA batteries (3), instruction manual, and instrument quality certificate packaged in a rugged thermoformed carrying case.
HI97769 is supplied with cuvettes (2), caps (2), plastic inserts for cuvettes (2), 1.5V AA batteries (3), instruction manual, and instrument quality certificate.
















Reviews
There are no reviews yet.