Importance of determination of Hardness and Alkalinity of water
With rise in the requirement of water for industrial, agricultural, and domestic consumption, resources are being used and re-used more
End of Content.
End of Content.

Among centuries of adaptation and development, the act of cleansing hair has changed quite dramatically; at first, a combination of oils, herbs, and perfumes was used as a way to simply freshen hair. Upon further understanding of hygienic health, soap was included in the combination to physically clean the hair. As the science behind hair care progressed, manufacturers synthesized chemical additives for use in shampoo as fragrances, foaming agents, and coloured dyes. Shampoo is now created and structured towards various hair types, as well as to achieve a certain end result, such as the reduction of dandruff. Both the synthetic and natural ingredients used in a shampoo dictate the pH of that particular product. The pH of a shampoo will alter the natural pH of skin and hair, which ideally falls between pH 3 and 5 and pH 4 and 5, respectively, thereby affecting their physical and chemical makeup.
The human scalp contains sebaceous glands which secrete sebum, a semiliquid substance composed of glycerides, waxes, and fatty acids. Sebum coats the outer layer of hair, called the cuticle, to prevent loss of water. The presence of sebum helps to maintain soft and flexible hair, as well as prevent the growth of bacteria and the spread of fungal infections on the scalp. However, due to its chemical makeup, sebum also attracts dirt. The shampoo is composed primarily of cleaning agents, such as detergents, that work to remove dirt and excess sebum, leaving a light layer of sebum on the scalp. A detergent molecule is composed of both nonpolar and polar portions, permitting oil and grease to be stripped from the scalp and hair by the nonpolar portion and washed away with water by the polar component; upon reaction with water, detergent molecules tend to produce alkaline solutions. Hair is composed of long, parallel chains of amino acids that are connected by forces such as hydrogen and disulphide bonds and salt bridges between acid and base groups. Environments that are either too alkaline or too acidic can affect and, at some levels, break these bonds. At pH levels between pH 1 and 2, both hydrogen bonds and salt bridges are broken. At slightly alkaline levels, closer to a pH of 8.5, some of the disulphide bonds are broken; with repeated washes at this pH, disulphide bonds will continue to break and result in “split ends.” Finally, at a pH near 12, all three types of bonds are broken and the hair dissolves. Ideally, shampoo should be “pH-balanced,” meaning that it would have a similar pH to that of hair to omit any negative effects on the scalp or hair due to pH variations. Some hair care companies produce pH-balanced shampoo, either with synthetic and/or natural components.
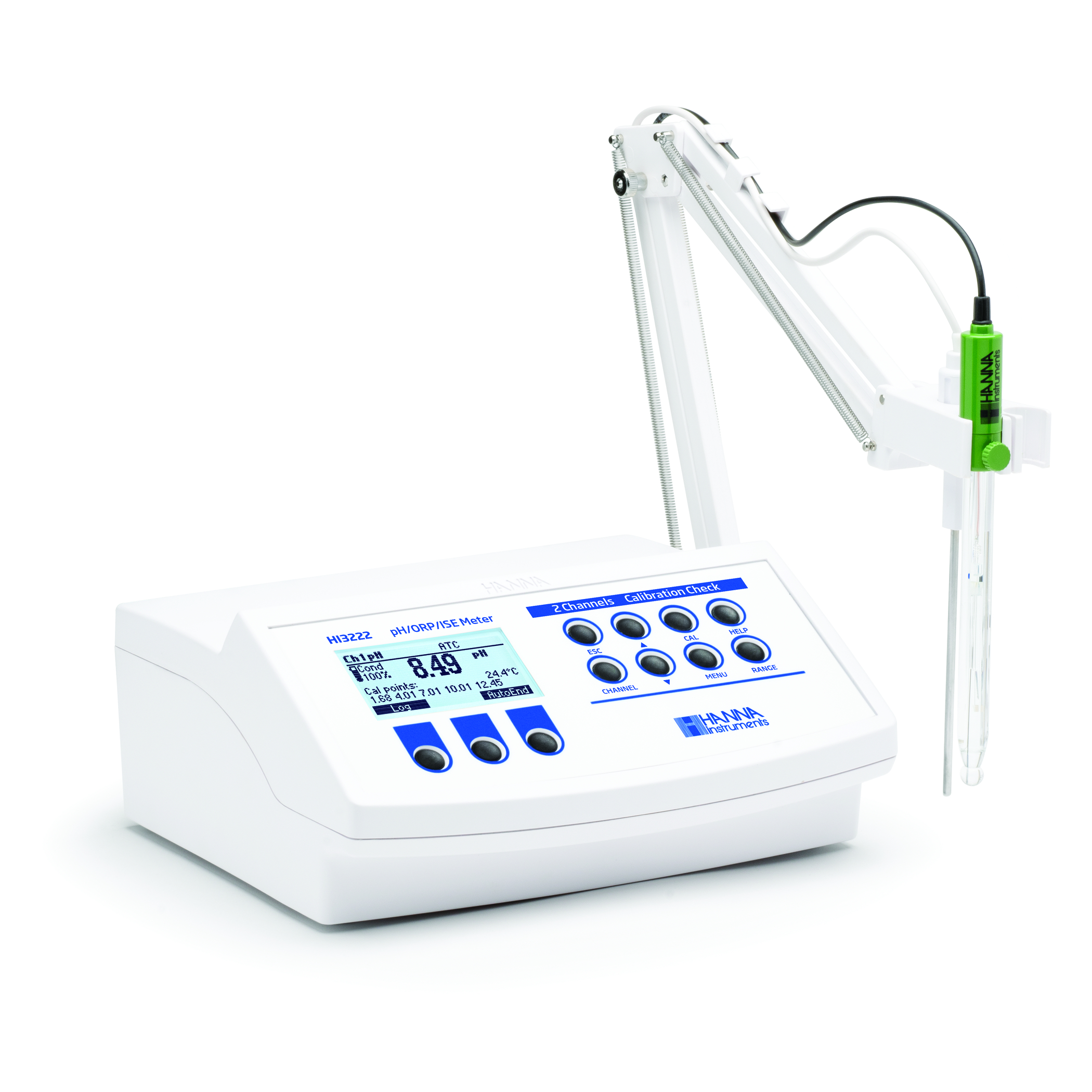
Hanna Instruments HI1053 pH Electrode for Fats and Creams is best for measuring pH in shampoo. The HI1053 provides a fast response time in the viscous sample due to the increased flow rate from outer reference that has a triple ceramic junction. A standard single ceramic junction in the outer reference has a flow rate of 15-20 μL/hour while the triple ceramic has a flow rate of 40-50 μL/hour. The HI1053 electrodes are available with different connection types.

With rise in the requirement of water for industrial, agricultural, and domestic consumption, resources are being used and re-used more
Any time you pick up a metal object, chances are that the item has some sort of plating on it.
To empower customers to achieve quality
by supplying intuitive, accurate, and reliable analytical instruments with exceptional customer service and value.
We take pride in every product we build. From an original idea, to a completed product ready for testing. We oversee every aspect of the manufacturing process. It is this level of attention to detail that sets us apart.
To empower customers to achieve quality by supplying intuitive, accurate, and reliable analytical instruments with exceptional customer service and value.
We take pride in every product we build. From an original idea, to a completed product ready for testing. We oversee every aspect of the manufacturing process. It is this level of attention to detail that sets us apart.
To empower customers to achieve quality by supplying intuitive, accurate, and reliable analytical instruments with exceptional customer service and value.
We take pride in every product we build. From an original idea, to a completed product ready for testing. We oversee every aspect of the manufacturing process. It is this level of attention to detail that sets us apart.