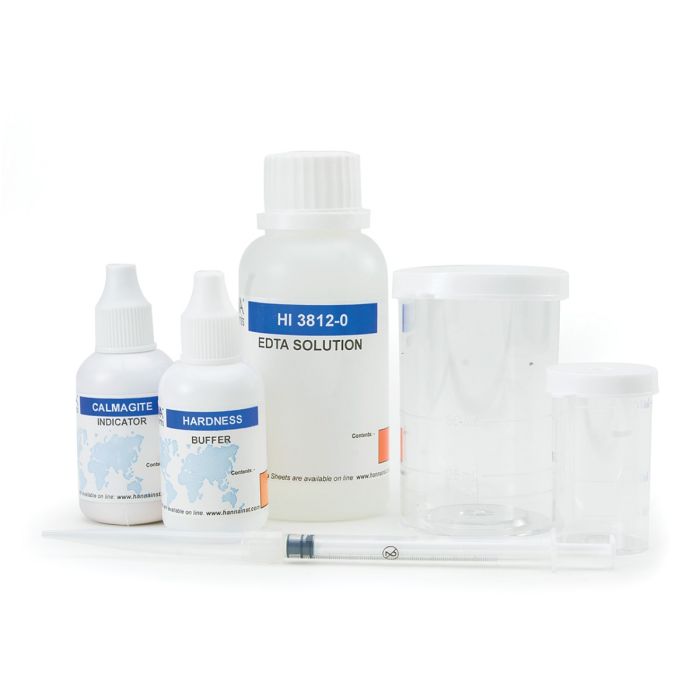
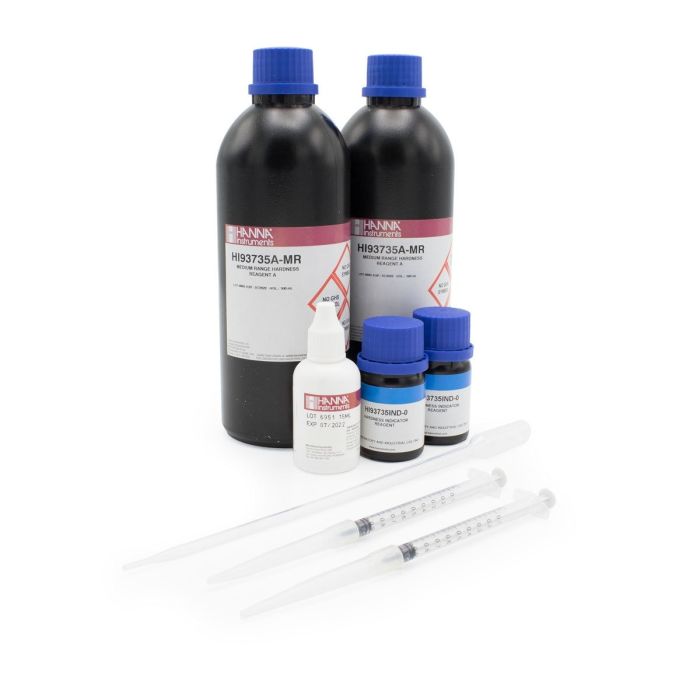
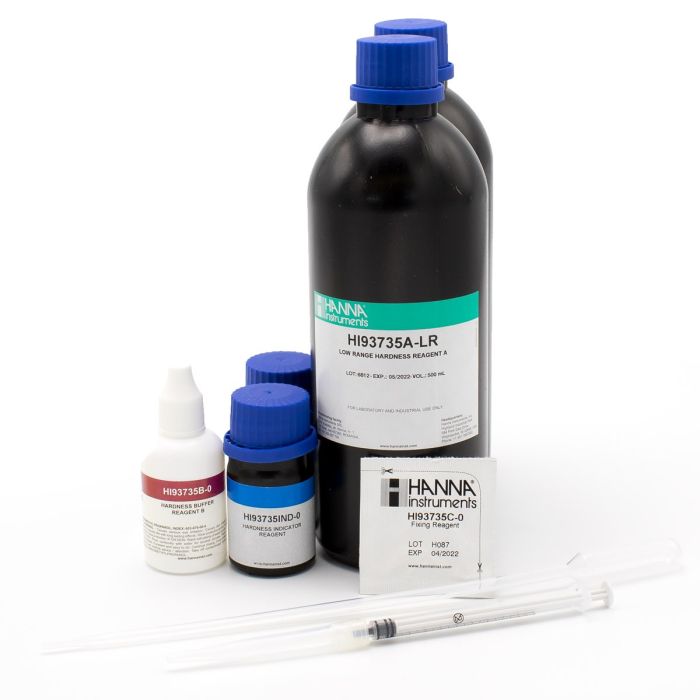
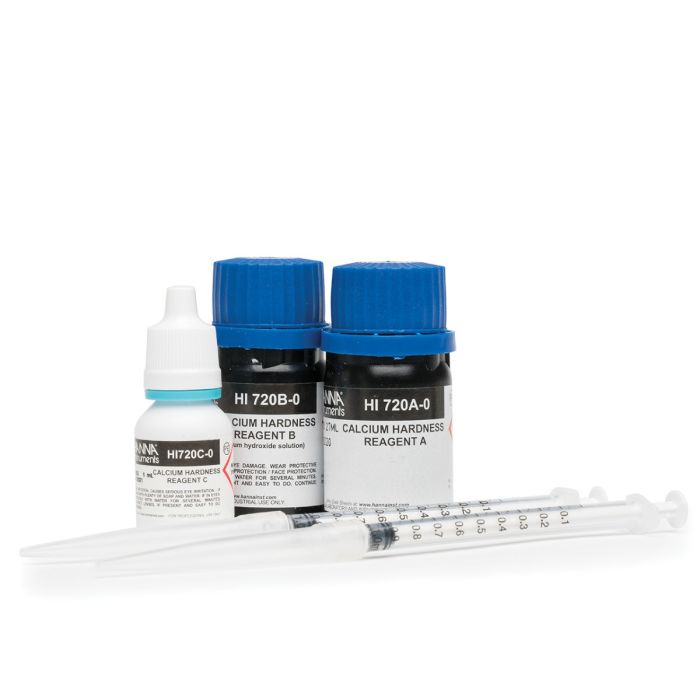
Hardness
Showing 1–20 of 25 results
End of Content.
End of Content.
Showing 1–20 of 25 results
To empower customers to achieve quality
by supplying intuitive, accurate, and reliable analytical instruments with exceptional customer service and value.
We take pride in every product we build. From an original idea, to a completed product ready for testing. We oversee every aspect of the manufacturing process. It is this level of attention to detail that sets us apart.
To empower customers to achieve quality by supplying intuitive, accurate, and reliable analytical instruments with exceptional customer service and value.
We take pride in every product we build. From an original idea, to a completed product ready for testing. We oversee every aspect of the manufacturing process. It is this level of attention to detail that sets us apart.
To empower customers to achieve quality by supplying intuitive, accurate, and reliable analytical instruments with exceptional customer service and value.
We take pride in every product we build. From an original idea, to a completed product ready for testing. We oversee every aspect of the manufacturing process. It is this level of attention to detail that sets us apart.